The Journal of Clinical and Preventive Cardiology has moved to a new website. You are currently visiting the old
website of the journal. To access the latest content, please visit www.jcpconline.org.
Original Article
Value of Ambulatory Blood Pressure Monitoring in Evaluation of Blood Pressure Control in Patients on Antihypertensive Treatment
Volume 1, Jul 2012
Manish Bansal, MD, DNB, Ravi R Kasliwal, MD, DM, Rahul Mehrotra, MD, DNB, Arun Rawat, MSc, Naresh Trehan, MD, Gurgaon, Haryana, India
J Clin Prev Cardiol 2012; 1(3):101-7
Hypertension is currently one of the leading killers worldwide. As per the World Health Organization estimates, suboptimal blood pressure (BP) is the most common attributable risk factor for death throughout the world. It is responsible for 62% of all cerebrovascular disease and 49% of all ischemic heart disease worldwide (1). Every 20 mmHg increase in systolic blood pressure (SBP) and every 10 mmHg increase in diastolic blood pressure (DBP) increases mortality from both ischemic heart disease and stroke by twofold (2). Consequently, effective control of BP remains the single-most effective therapeutic strategy to prevent cardiovascular morbidity and mortality. It is estimated that a 5 mmHg reduction in SBP in the population is likely to result in a 14% overall reduction in mortality due to stroke, a 9% reduction in mortality due to ischemic heart disease, and a 7% decrease in all-cause mortality (3,4).Conventionally, diagnosis and management of hypertension are based on office-based recordings of BP. However, it is increasingly recognized that a single office measurement of BP may not accurately reflect a patient’s actual BP burden. Studies using prolonged ambulatory BP (ABP) monitoring have revealed significant differences between the office BP measurements and the 24-h average BP measured by ambulatory recordings (5–7). Thus, the antihypertensive treatment based on office BP measurement alone may be misleading and inappropriate and ABP monitoring may prove to be useful in overcoming these limitations (6,8,9). However, as there is no data to document value of ABP monitoring in guiding antihypertensive therapy in Indian patients, we sought this study to derive information about potential utility of ABP monitoring in our patients receiving treatment for hypertension.
Methods
One hundred and fifty eight consecutive hypertensive subjects, who were on pharmacological treatment for hypertension and who had undergone ABP monitoring, were included in this retrospective study. The ABP monitoring was performed for clinical indications, as advised by their treating physicians. The monitoring was performed using the commercially available devices (Tonoport V, GE Medical Systems) for this purpose which employed oscillometric method for measuring BP. The typical recording duration was 24 h, with measurements repeated every 30 min during the day-time and every 60 min during the night-time.
One hundred and fifty eight consecutive hypertensive subjects, who were on pharmacological treatment for hypertension and who had undergone ABP monitoring, were included in this retrospective study. The ABP monitoring was performed for clinical indications, as advised by their treating physicians. The monitoring was performed using the commercially available devices (Tonoport V, GE Medical Systems) for this purpose which employed oscillometric method for measuring BP. The typical recording duration was 24 h, with measurements repeated every 30 min during the day-time and every 60 min during the night-time.
The patients’ age, gender, office BP recordings and findings on ABP monitoring were retrieved from the ABP monitoring lab records. The patients were divided in two groups, based on their office BP recordings. If the office BP values were <140/90 mmHg, the patients were considered to have “controlled office BP,” whereas those with higher values were labeled as having “uncontrolled office BP.”
On ABP monitoring, the average SBP and DBP over the entire monitoring period, during the day-time and during the night-time were recorded. The ABP was considered abnormal if the average values were >140/90 mmHg during the day-time or >135/85 mmHg over the entire monitoring period or >125/75 mmHg during the nighttime (10). The percentage of BP recordings above these cut-off values were also noted. Day-time BP load was considered to be increased if at least 40% of all day-time SBP or DBP values were above the cut-off limits (11). In addition, the nocturnal drop in SBP and DBP was also recorded. A nocturnal drop of at least 10% from day-time average SBP or DBP was considered to be normal and such patients were labeled as “dippers” (10). Those who had less marked fall in BP were labeled as nondippers, whereas those with paradoxical rise, instead of normal fall in BP during night-time, were labeled as inversedippers.
Statistical Analysis
The data was managed on Microsoft excel spreadsheet (version 2007, Microsoft Corp, Seattle, WA). Values were expressed as mean (± standard deviation) or as percentages. Comparisons between the groups were done using Student’s unpaired t-test or chi-square test wherever appropriate. A p value <0.05 was considered statistically significant. Statistical analysis was performed using SPSS for Windows (release 15.0, SPSS Inc).
Results
The mean age of the subjects was 50.5 ± 16.1 years and 74.7% of them were males. Overall, the mean office SBP and DBP were 140.9 ± 18.6 mmHg and 89.1 ± 13.4 mmHg, respectively. Of the 158 subjects, 62 (39.2%) had “controlled office BP” (office BP <140/90 mmHg) and the remaining 96 patients had “uncontrolled office BP” (office BP > 140 mmHg systolic and/or > 90 mmHg diastolic). There was no difference between the two groups with respect to age and gender (Table 1).
.jpg )
The values are mean ± standard deviation or actual numbers with percentages in parentheses. BP, blood pressure.
.jpg )
The values are mean ± standard deviation or actual numbers with percentages in parentheses. BP, blood pressure.
ABP Monitoring
Overall, the ABP monitoring was performed for an average of 25.7 ± 7.3 h, which included 15.7 ± 4.5 h of day-time recordings and 9.9± 3.1 h of night-time recordings.
Average BP values and BP load
(Table 2, Figures 1 and 2)
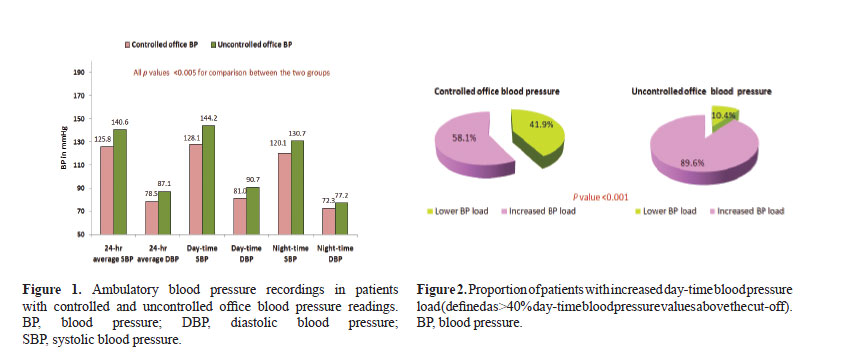
As compared to the patients with uncontrolled office BP, those with controlled office BP had lower average 24-h ABP (SBP 125.8 ± 9.8 vs. 140.6 ± 13.3 mmHg, DBP 78.5 ± 8.0 vs. 87.1 ± 9.7 mmHg, p value <0.001 for both), day-time ABP (SBP 128.1 ± 9.0 vs. 144.2 ± 13.2 mmHg, DBP 81.0 ± 8.6 vs. 90.7 ± 9.9 mmHg, p value <0.001 for both) and night-time ABP (SBP 120.1 ± 14.0 vs. 130.7 ± 16.0 mmHg, p value <0.001; DBP 72.3 ± 9.9 vs. 77.2 ± 10.8 mmHg, p value 0.005). Overall, 24.2% patients with controlled office BP had average ABP in the abnormal range as compared to 72.9% patients with uncontrolled office BP (p <0.001) (Table 2, Fig. 1).
Of the 62 patients with controlled office BP, 26 (41.9%) had at least 40% of the day-time BP values above the normal range and thus had increased day-time BP load. In contrast, nearly 90% of the patients with uncontrolled office BP had increased day-time BP load (p <0.001) (Fig. 2).
.jpg )
.jpg )
Discussion

Nocturnal fall in BP (Table 2, Figures 3)
The patients with controlled office BP had less marked drop in BP during night-time as compared to the patients with uncontrolled office BP (SBP 10.5 ± 10.7% vs. 14.4 ± 9.0%, p value 0.013; DBP 6.3 ± 7.8% vs. 9.3 ± 7.0%, p value 0.014). More than half of the patients in both the groups (54.8% of those with controlled office BP and 52.1% with uncontrolled office BP, p - NS) were classified as nondippers based on SBP reduction during the night-time. The fall in DBP was much greater with only less than one-third of the patients being nondippers. However, more patients with controlled office BP (32.3%) were nondippers as compared to those with uncontrolled office BP (18.8%, p value = 0.014). In addition, a significant proportion of patients had paradoxical night-time rise in SBP or DBP, which again was more common in patients with controlled office BP (21.0% vs. 10.4% with, p = 0.066).
.jpg )
.jpg )
Discussion
In the present study, we investigated the role of ABP monitoring in assessing the adequacy of BP controlin hypertensive subjects receiving antihypertensive treatment. The study showed that almost a quarter of all patients with controlled office BP had elevated average BP on 24-h ABP monitoring. Similarly, a quarter of those with uncontrolled office BP had normal average BP on ABP monitoring. Thus, ABP monitoring could potentially alter treatment in almost 25% of all patients. In addition, a significant proportion of the patients in both the groups did not have adequate drop in blood pressure during night-time – a finding which was paradoxically more common in patients with controlled office BP.
It is well known that BP is controlled by a number of neurohormonal factors, which are responsible for the significant temporal variability seen in BP during normal life as well as in disease states. Accordingly, a single BP reading, as obtained in a physician’s office, is unlikely to reflect the overall status of BP burden in a given individual (6,7). In contrast, ABP monitoring allows prolonged BP monitoring in a setting which is very close to the patients’ daily life and is therefore believed to provide more accurate estimates of true BP status and the resultant vascular injury. Indeed, a number of studies have shown that compared to the office BP, ABP predicts cardiovascular risk better and has much stronger correlation with hypertension-related organ damage (12–19). Furthermore, ABP has been shown to provide more accurate estimates of BP reduction with antihypertensive therapy and is more accurate in tracking improvements in end-organ structure and function achieved with effective BP control (19–23). Given these advantages, ABP monitoring has been recommended as a routine modality for diagnosis of hypertension as well as for monitoring response to antihypertensive treatment (9).
Numerous studies have demonstrated the utility of ABP monitoring in guiding antihypertensive management in clinical practice and in comparing BP-lowering efficacy of different pharmacological agents (8,20,23–25). In a study comparing placebo and an alpha-adrenergic blocker terazosin, Drayer et al. showed that ABP was more sensitive in detecting changes in BP with tearzosin than office BP measurements (23). In a randomized, controlled study involving 419 patients, Staessen et al. showed that treatment guided by ABP monitoring resulted in much lower requirement of antihypertensive drugs without adversely affecting BP control, general well-being and end-organ damage (20). In our study, we found that almost a quarter of the patients with controlled office BP actually had increased 24-h average BP on ABP monitoring and were therefore candidates for more intensive treatment. At the same time, more than a quarter of all the patients with uncontrolled office BP had normal 24-h average BP. After excluding from these the 16.7% patients who had increased day-time BP load, almost 10% patients still remained who had reasonably well-controlled BP and therefore did not require any further intensification of antihypertensive therapy. Thus, ABP monitoring in our study could alter treatment in almost a quarter of all the patients, with significant implications. On one hand, it could protect patients from the potential ill-effects of inadequate BP control and on the other hand, it could save patients from the harmful side-effects and the cost of the unnecessary dditional treatment.
An added advantage of ABP monitoring is its ability to allow assessment of diurnal variations of BP which have important prognostic implications. In normotensive subjects, BP has a distinct circadian pattern. It tends to increase during day-time, peaks during the day and then falls to a nadir at midnight, before rising again early in the morning. Both, a lack of adequate drop in BP during night-time and an exaggerated early morning surge are associated with adverse outcomes (26–29). Night-time BP, in particular, has been shown to be an important determinant of outcomes in patients with hypertension. There is evidence to suggest that the night-time BP may even be superior to day-time BP in prediction of cardiovascular risk (15–18). In addition, subjects in whom nocturnal decrease in BP is blunted (nondippers) or those who have paradoxical rise in BP have been reported to have a greater prevalence of organ damage and a greater risk of adverse cardiovascular events (26–28,30,31). In a recent study involving 30 Indian subjects, Pai et al. found that night BP measurements were significantly lower in subjects without left ventricular hypertrophy and had statistically significant correlation with the left ventricular mass index (31). In our study, we found that a significant proportion of patients did not have adequate drop in BP or actually had paradoxical rise in BP during night-time which would indicate increased long-term cardiovascular risk in these subjects. However, it can be argued that the antihypertensive therapy itself could have blunted nocturnal drop in BP as evidenced by lower day–night difference in BP in patients with controlled office BP as compared to those with uncontrolled office BP. Nonetheless, paradoxical rise in night-time BP, which was seen in as many as 15% of all subjects, was clearly an abnormal finding and could not have resulted from the antihypertensive treatment.
Limitations
The study had some important limitations that need to be considered. Due to its retrospective nature, we could not obtain detailed information about the baseline clinical and biochemical parameters of the patients and hence could not determine their influence on BP control. However, the lack of this data did not affect the primary purpose of the study which was to observe the differences between the office measurements of BP and the ABP monitoring. Similarly, we could not ascertain the effect of different antihypertensive agents on BP control, particularly the magnitude and duration of BP reduction with different agents. However, as mentioned above, this too did not compromise the primary aim of the study.
Conflict of Interest
Acknowledgement
Conclusions
Although, compared to the patients with uncontrolled office BP, those with controlled office BP had lower BP readings on ABP monitoring, a substantial proportion of them still had uncontrolled 24-h average BP and increased overall BP load. At the same time, many of the patients with uncontrolled office BP had normal average BP on ABP monitoring. Thus, ABP monitoring could potentially alter treatment in almost 25% of all patients. In addition, ABP monitoring also disclosed blunted or even reversed nocturnal drop in BP in a significant number of these patients. The significance of these findings in day-to-day management of hypertension needs to be assessed in large, prospective studies.
Conflict of Interest
None
Acknowledgement
We sincerely thank Mr Ashish Rawat, Technician, Noninvasive Cardiology, for his help in data collection and compilation.
References
References
- R Guilbert JJ. The world health report 2002 – reducing risks, promoting healthy life. Educ Health (Abingdon). 2003; 16:230.
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002; 360:1903–13.
- Whelton PK, He J, Appel LJ, Cutler JA, Havas S, Kotchen TA, Roccellac EJ, Stout R, Vallbona C, Winston MC, Karimbakas J. Primary prevention of hypertension: clinical and public health advisory from The National High Blood Pressure Education Program. JAMA 2002; 288:1882–8.
- Stamler R. Implications of the INTERSALT study. Hypertension. 1991; 17:I16–20.
- Little P, Barnett J, Barnsley L, Marjoram J, Fitzgerald-Barron A, Mant D. Comparison of agreement between different measures of blood pressure in primary care and daytime ambulatory blood pressure. BMJ 2002; 325:254.
- Banegas JR, Segura J, Sobrino J, Rodriguez-Artalejo F, de la Sierra A, de la Cruz JJ, Gorostidi M, Sarria A, Ruilope LM. Effectiveness of blood pressure control outside the medical setting. Hypertension. 2007; 49:62–8
- Gorostidi M, Sobrino J, Segura J, Sierra C, de la Sierra A, Hernandez del Rey R, Vinyoles E, Galceran JM, Lopez-Eady MD, Marin R, Banegas JR, Sarria A, Coca A, Ruilope LM. Ambulatory blood pressure monitoring in hypertensive patients with high cardiovascular risk: a cross-sectional analysis of a 20,000-patient database in Spain. J Hypertens. 2007; 25:977–84.
- Uallachain GN, Murphy G, Avalos G. The RAMBLER study: the role of ambulatory blood pressure measurement in routine clinical practice: a cross-sectional study. Ir Med J. 2006; 99:276–9.
- Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, Grassi G, Heagerty AM, Kjeldsen SE, Laurent S, Narkiewicz K, Ruilope L, Rynkiewicz A, Schmieder RE, Struijker Boudier HA, Zanchetti A, Vahanian A, Camm J, De Caterina R, Dean V, Dickstein K, Filippatos G, Funck-Brentano C, Hellemans I, Kristensen SD, McGregor K, Sechtem U, Silber S, Tendera M, Widimsky P, Zamorano JL, Kjeldsen SE, Erdine S, Narkiewicz K, Kiowski W, Agabiti-Rosei E, Ambrosioni E, Cifkova R, Dominiczak A, Fagard R, Heagerty AM, Laurent S, Lindholm LH, Mancia G, Manolis A, Nilsson PM, Redon J, Schmieder RE, Struijker-Boudier HA, Viigimaa M, Filippatos G, Adamopoulos S, Agabiti-Rosei E, Ambrosioni E, Bertomeu V, Clement D, Erdine S, Farsang C, Gaita D, Kiowski W, Lip G, Mallion JM, Manolis AJ, Nilsson PM, O’Brien E, Ponikowski P, Redon J, Ruschitzka F, Tamargo J, van Zwieten P, Viigimaa M, Waeber B, Williams B, Zamorano JL, The task force for the management of arterial hypertension of the European Society of H, The task force for the management of arterial hypertension of the European Society of C. 2007 Guidelines for the management of arterial hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J. 2007; 28:1462–536.
- Pickering TG, Hall JE, Appel LJ, Falkner BE, Graves J, Hill MN, Jones DW, Kurtz T, Sheps SG, Roccella EJ. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation. 2005; 111:697–716.
- White WB, Dey HM, Schulman P. Assessment of the daily blood pressure load as a determinant of cardiac function in patients with mildto- moderate hypertension. Am Heart J. 1989; 118:782–95.
- Clement DL, De Buyzere ML, De Bacquer DA, de Leeuw PW, Duprez DA, Fagard RH, Gheeraert PJ, Missault LH, Braun JJ, Six RO, Van Der Niepen P, O’Brien E. Prognostic value of ambulatory blood-pressure recordings in patients with treated hypertension. N Engl J Med. 2003; 348:2407–15.
- Imai Y, Ohkubo T, Sakuma M, Tsuji II, Satoh H, Nagai K, Hisamichi S, Abe K. Predictive power of screening blood pressure, ambulatory blood pressure and blood pressure measured at home for overall and cardiovascular mortality: a prospective observation in a cohort from Ohasama, northern Japan. Blood Press Monit. 1996; 1:251–4.
- Mancia G, Parati G, Hennig M, Flatau B, Omboni S, Glavina F, Costa B, Scherz R, Bond G, Zanchetti A. Relation between blood pressure variability and carotid artery damage in hypertension: baseline data from the European Lacidipine Study on Atherosclerosis (ELSA). J Hypertens 2001; 19:1981–9.
- Staessen JA, Thijs L, Fagard R, O’Brien ET, Clement D, de Leeuw PW, Mancia G, Nachev C, Palatini P, Parati G, Tuomilehto J, Webster J. Predicting cardiovascular risk using conventional vs ambulatory lood pressure in older patients with systolic hypertension. Systolic Hypertension in Europe Trial Investigators. JAMA 1999; 282:539–46.
- Sega R, Facchetti R, Bombelli M, Cesana G, Corrao G, Grassi G, Mancia G. Prognostic value of ambulatory and home blood pressures compared with office blood pressure in the general population: followup results from the Pressioni Arteriose Monitorate e Loro Associazioni (PAMELA) study. Circulation. 2005; 111:1777–83.
- Dolan E, Stanton A, Thijs L, Hinedi K, Atkins N, McClory S, Den Hond E, McCormack P, Staessen JA, O’Brien E. Superiority of ambulatory over clinic blood pressure measurement in predicting mortality: the Dublin outcome study. Hypertension. 2005; 46:156–61.
- Kikuya M, Ohkubo T, Asayama K, Metoki H, Obara T, Saito S, Hashimoto J, Totsune K, Hoshi H, Satoh H, Imai Y. Ambulatory blood pressure and 10-year risk of cardiovascular and noncardiovascular mortality: the Ohasama study. Hypertension. 2005; 45:240–5.
- Verdecchia P, Schillaci G, Guerrieri M, Gatteschi C, Benemio G, Boldrini F, Porcellati C. Circadian blood pressure changes and left ventricular hypertrophy in essential hypertension. Circulation. 1990; 81:528–36.
- Staessen JA, Byttebier G, Buntinx F, Celis H, O’Brien ET, Fagard R. Antihypertensive treatment based on conventional or ambulatory blood pressure measurement. A randomized controlled trial. Ambulatory Blood Pressure Monitoring and Treatment of Hypertension Investigators. JAMA 1997; 278:1065–72.
- Mancia G, Zanchetti A, Agabiti-Rosei E, Benemio G, De Cesaris R, Fogari R, Pessina A, Porcellati C, Rappelli A, Salvetti A, Trimarco B. Ambulatory blood pressure is superior to clinic blood pressure in predicting treatment-induced regression of left ventricular hypertrophy. SAMPLE Study Group. Study on Ambulatory Monitoring of Blood Pressure and Lisinopril Evaluation. Circulation. 1997; 95:1464–70.
- Fagard RH, Staessen JA, Thijs L. Relationships between changes in left ventricular mass and in clinic and ambulatory blood pressure in response to antihypertensive therapy. J Hypertens. 1997; 15:1493–502.
- Drayer JI, Weber MA, DeYoung JL, Brewer DD. Long-term BP monitoring in the evaluation of antihypertensive therapy. Arch Intern Med. 1983; 143:898–901.
- Mancia G, Omboni S, Ravogli A, Parati G, Zanchetti A. Ambulatory blood pressure monitoring in the evaluation of antihypertensive treatment: additional information from a large data base. Blood Press. 1995; 4:148–56.
- Smith DH, Dubiel R, Jones M. Use of 24-hour ambulatory blood pressure monitoring to assess antihypertensive efficacy: a comparison of olmesartan medoxomil, losartan potassium, valsartan, and irbesartan. Am J Cardiovasc Drugs. 2005; 5:41–50.
- Liu M, Takahashi H, Morita Y, Maruyama S, Mizuno M, Yuzawa Y, Watanabe M, Toriyama T, Kawahara H, Matsuo S. Non-dipping is a potent predictor of cardiovascular mortality and is associated with autonomic dysfunction in haemodialysis patients. Nephrol Dial Transplant. 2003; 18:563–9.
- Cicconetti P, Morelli S, Ottaviani L, Chiarotti F, De Serra C, De Marzio P, Costarella M, Sgreccia A, Ciotti V, Marigliano V. Blunted nocturnal fall in blood pressure and left ventricular mass in elderly individuals with recently diagnosed isolated systolic hypertension. Am J Hypertens. 2003; 16:900–5.
- Tseng YZ, Tseng CD, Lo HM, Chiang FT, Hsu KL. Characteristic abnormal findings of ambulatory blood pressure indicative of hypertensive target organ complications. Eur Heart J. 1994; 15:1037– 43.
- Kario K, Pickering TG, Umeda Y, Hoshide S, Hoshide Y, Morinari M, Murata M, Kuroda T, Schwartz JE, Shimada K. Morning surge in blood pressure as a predictor of silent and clinical cerebrovascular disease in elderly hypertensives: a prospective study. Circulation. 2003; 107:1401–6.
- O’Brien E, Sheridan J, O’Malley K. Dippers and non-dippers. Lancet. 1988; 2:397.
- Pai AU, Chakrapani M, Bhaskaran U, Kamath P. Study of homemonitored night blood pressure and its correlation with left ventricular hypertrophy in treatment-naive hypertensive patients. Singapore Med J. 2012; 53:95–8.
- Why Publish with JCPC?
- Instructions to the Authors
- Submit Manuscript
- Advertise with Us
- Journal Scientific Committee
- Editorial Policy
Print: ISSN: 2250-3528


